Our Process
How Do Your Legs Feel & Look Right Now?

STEP 1: Consultation
At Vein & Vascular Institute, you will be listened to and encounter a friendly staff that will make sure you get the best care.
Care will come from his team of experts which includes a provider and an ultrasonographer on the first visit.
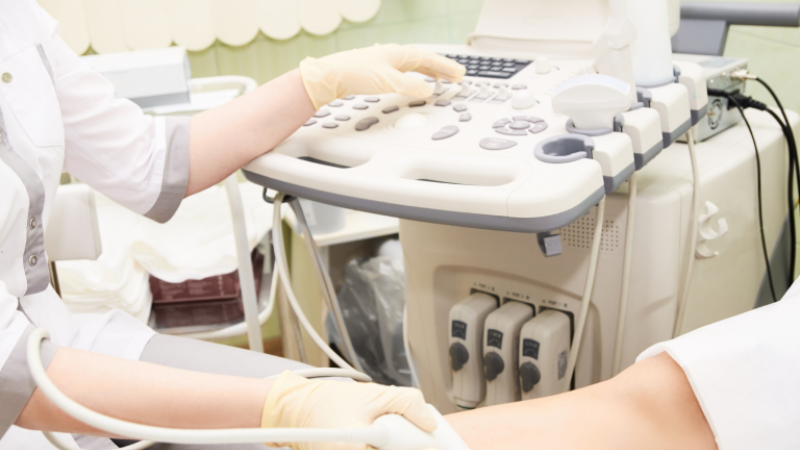
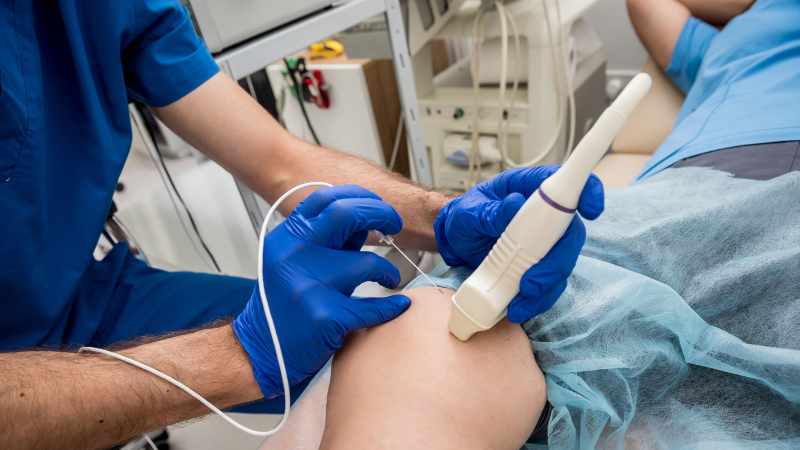
STEP 2: Ultrasound
You won’t do that at Vein & Vascular Institute.
We do an ultrasound and diagnostics onsite at our IAC credentialled Vascular Testing Lab to ensure your time is not wasted. You will get same-day results. You will get same-day results from one of our Registered Vascular Technologists.
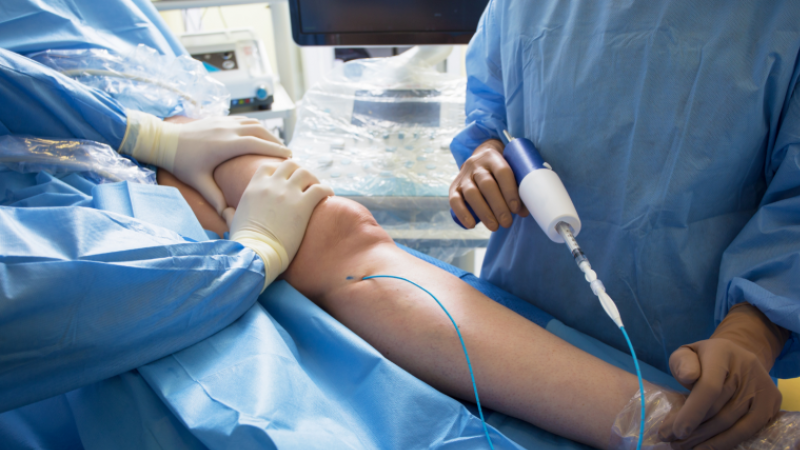
STEP 3: Treatment Plan
Sclerotherapy
The sclerosant placed in the vein causes little to no pain during treatment and is overall tolerated much better than laser therapy. The process causes a gradual fading of the treated vessel over a period of several weeks to several months.
Click here to read an in-depth article on Sclerotherapy authored by Dr. Dietzek.
Ultrasound Guided Sclerotherapy
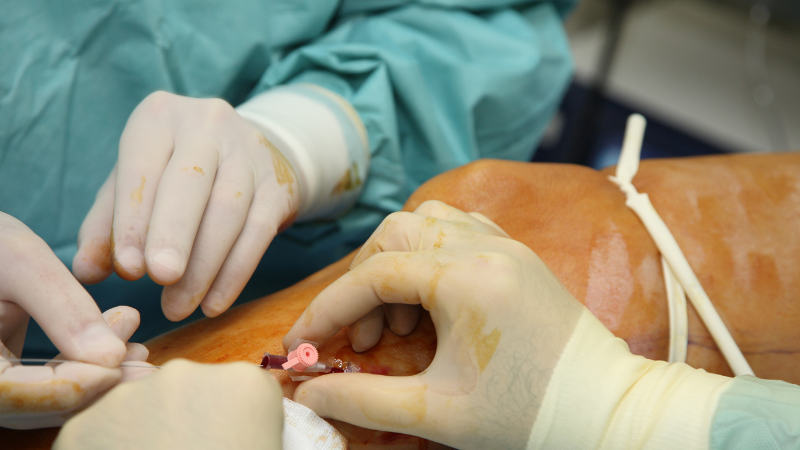
Microphlebectomy
Closurefast™
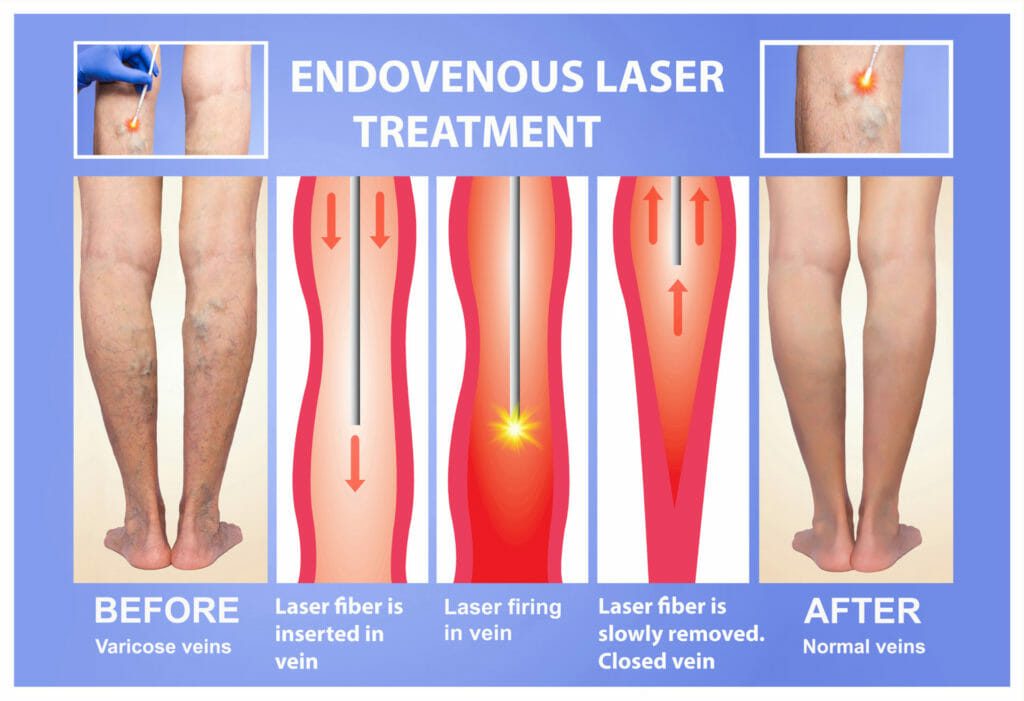
After the procedure, blood is naturally re-routed through other, healthier veins. The procedure’s benefits include faster treatment and recovery time, less discomfort during and after, and little or no scarring, bruising, or swelling. ClosureFast™ is performed under local anesthesia in as little as 20-30 minutes. Typically patients return to their normal activities in less than 24 hours.

Clarivein®
ClariVein®IC is a specialty infusion catheter with a rotating wire tip designed for the controlled 360-degree dispersion of physician-specified agents to the targeted treatment area.
The ClariVein®IC device is a thin catheter (tube) that your doctor temporarily inserts into the peripheral vasculature through a pin-sized entrance point. ClariVein®IC is several times smaller than other devices used in peripheral vascular treatments allowing the entrance point to be smaller.
Another benefit of ClariVein is Use of the it does NOT require painful anesthesia and will not cause thermal agitation.
Venaseal™


Microfoam
Microfoam injection is a minimally invasive vein treatment for spider veins and varicose veins. This non-invasive, outpatient procedure uses an injectable foam solution.
During the treatment, the doctor will administer a small amount of microfoam. The microfoam fills the desired section of the vein and causes the diseased vein to collapse.
Blood flow is redirected to healthier nearby veins. The microfoam disperses as it comes into contact with blood in healthy veins.
Endovenous Laser Therapy (EVLT)
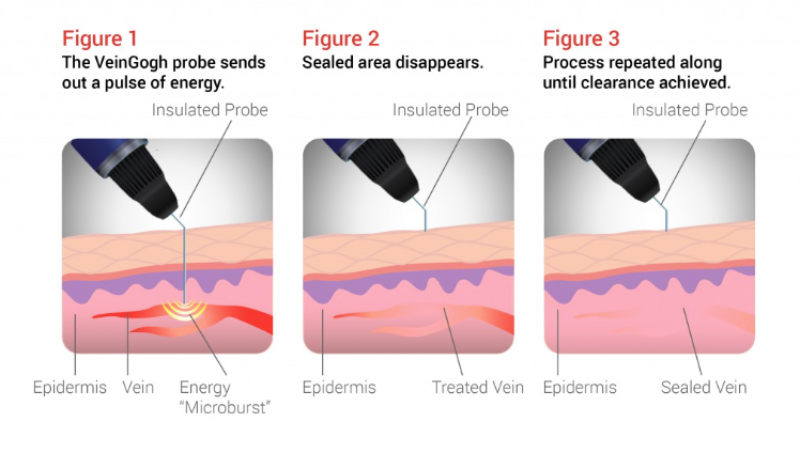
VeinGogh
Laser Ablation of Spider Veins

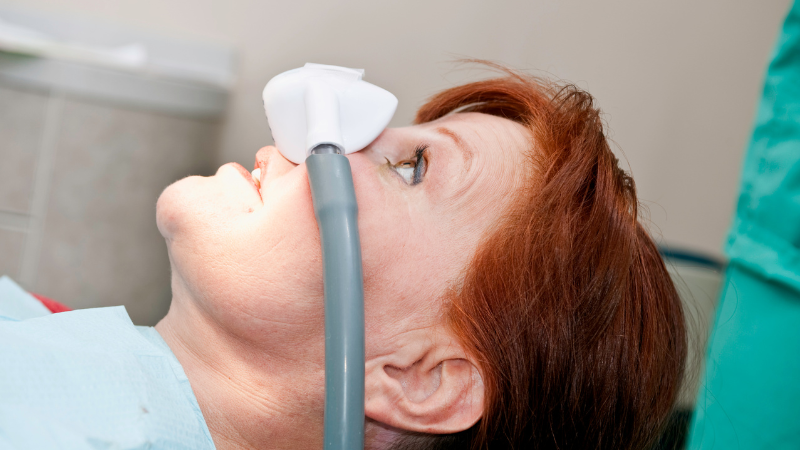
Nitronox Plus®
FAQs
Veins are the vessels that return blood to the heart once it has circulated through the body (as opposed to arteries, which carry oxygen-rich blood from the heart to the body). They have one-way valves that help keep blood flowing in the proper direction. If these valves stop functioning the way they are supposed to, blood can flow backwards and pool in the vein, causing it to stretch. These enlarged blood vessels are classified into two groups: spider veins and varicose veins.
Spider veins are visible red or blue blood vessels that may spread like a web across the skin anywhere on the body, most commonly on the face and legs. Varicose veins are blood vessels with weak walls that swell or balloon outward, raising the skin surface.
Yes. More than 80 million people in the United States have problems with their veins, including about 50 percent of women.
It is not entirely known what causes spider and varicose veins, although many likely factors have been identified. The most significant contributing factor is heredity. Others include:
- Puberty
- Pregnancy
- Menopause
- Birth control pills
- Estrogen and progesterone
- Aging
- Occupations that involve standing
- Obesity
- Leg Injury
Yes. Abnormal veins in the legs can cause pain (including fatigue, heaviness, aching, burning, throbbing, itching, cramping and restlessness) and prevent proper skin nutrition, leading to eczema, inflammation and ulceration.
Venous insufficiency is when the vein valves are not working correctly to bring blood back to the heart. The valve dysfunction allows blood to flow in the wrong direction. There are three types. Superficial, perforator and deep insufficiency. In deep and superficial insufficiency the blood flows down the leg. With perforator insufficiency the blood from the deep veins within the muscles flows towards the superficial network of veins. An ultrasound study is utilized to evaluate for all three types.
We first perform a complete history and physical examination. While a patient may note only dilated veins and spider veins on the lower leg, it is quite common that vein valve dysfunction originating higher in the leg at the groin may be the cause for their condition.
The most effective and reliable method to determine both the precise areas of vein valve dysfunction is an ultrasound examination of the entire lower leg. A duplex scan (ultrasound) enables us to look at the roadmap of the vein as well as the direction of blood flow and valve function.
Our vascular labs, conveniently located at our two office locations, is very experienced in examining a wide variety of vein problems. Ultrasound gives highly reliable results so that we can offer the best treatment plan. The test is painless and uses a gel on the skin to allow imaging of the veins underneath the skin and deep in the leg. The tests are done with the leg vein valves placed in the position of stress which is with the patient standing. You should bring a pair of loose fitting shorts for your test.
Venefit™ is used to treat reflux most commonly in the superficial system and as needed in perforator veins. The superficial system is comprised of the greater and small saphenous veins.
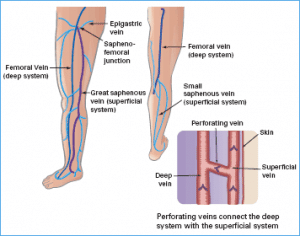
The varicose veins you see are typically swollen stretched branches from the superficial system, like branches off a tree trunk. When the superficial refluxing vein (the trunk) is closed, the flow of blood into the varicose branches is substantially reduced. The varicose veins will typically decrease in size. In some patients the varicose veins will no longer be noticeable. In other patients the veins may still be present but smaller. These can typically be addressed with either sclerotherapy or microphlebectomy.
This is a neurologic condition where there is constant uncontrolled movement of the legs typically during the night. Similar symptoms however can occur with venous insufficiency (reflux) leading to misdiagnosis.
With venous reflux, veins stretch causing swelling and pain (aching, heaviness). Frequently patients also complain of cramping at bedtime, crawling skin sensation, itching and restless legs.
If you have varicose veins or leg swelling, be certain you are evaluated by a vein specialist before being treated with medication for “Restless Leg Syndrome”. When the abnormal veins are treated, these symptoms resolve and no prescription medication is needed.
Yes. Hand vein therapy is available as a cosmetic procedure and can improve the appearance of bulging veins that become more prominent as our skin thins with age. The treatment may vary by individual and may include sclerotherapy. Mild hand swelling occurs for a few days, but recovery time is generally short with no restriction on activities.
Yes. Physical examination, often with the aid of specialized equipment such as non-invasive ultrasound, by a professional can detect problems with blood vessels that aren’t visible otherwise.
This is a neurologic condition where there is constant uncontrolled movement of the legs typically during the night. Similar symptoms however can occur with venous insufficiency (reflux) leading to misdiagnosis.
With venous reflux, veins stretch causing swelling and pain (aching, heaviness). Frequently patients also complain of cramping at bedtime, crawling skin sensation, itching and restless legs.
If you have varicose veins or leg swelling, be certain you are evaluated by a vein specialist before being treated with medication for “Restless Leg Syndrome”. When the abnormal veins are treated, these symptoms resolve and no prescription medication is needed.
Detergent type of sclerosants can be foamed. Air and the sclerosant solution is mixed in varying concentrations to produce a foam. When injected into the vein it dwells for a longer period of time in the vein. This results in greater irritation with a lower concentration of sclerosant. It can enhance results and minimize complications.
The name sounds funny but it is called Horse Chestnut Seed extract. It comes in topical or pill form. It is thought to be a mild diuretic and vasoconstrictor. This may improve the appearance of spider veins. It does not address the underlying problem.
Yes. Spider veins and varicose vein formation and complications may be prevented by wearing sunscreen on the face, exercising regularly, controlling your weight, elevating legs when resting, not crossing the legs when sitting, avoiding long periods of standing, wearing elastic support stockings, and eating high-fiber foods.
Yes, when administered by a trained professional in a controlled setting, Nitronox Plus® is perfectly safe. If you have doubts about your ability to drive safely after receiving laughing gas, it’s best to arrange alternative transportation.
